- NRX is focused on treating central nervous system injuries, spinal cord injury (SCI), optic nerve injury, brain trauma, and neurological conditions through regenerative medicine. Its lead product, ExoPTEN, is an exosome-based therapy that has completed several preclinical studies, and received an orphan drug designation from the U.S. FDA, and the European Medicines Agency (EMA), for the treatment of acute SCI. While there is currently no cure for SCI, treatments such as immobilization, surgery, medication, and rehabilitation can help manage symptoms.
- The company has also conducted a pre-Investigational New Drug (pre-IND) meeting with the FDA, a necessary step before initiating human clinical trials. ExoPTEN is expected to enter phase one clinical trials by the end of 2025.
- We note that the common exit strategy for pharma/biotech companies is to either be acquired by larger companies, or enter into licensing agreements with them, following promising clinical trial results.
Risks
- Limited operating history
- In pre-revenue stage
- No guarantee that any of its drugs/therapies will be commercialized
- Potential for delays in clinical trials; unfavorable results
- Will need to pursue equity financings, implying potential for share dilution
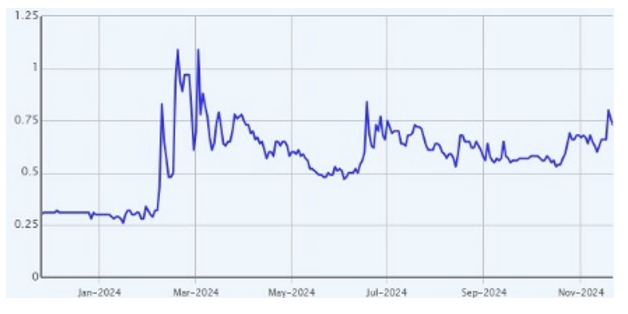
Price Performance (YTD)
| |
YTD |
12M |
| NRX |
157% |
148% |
| TSXV |
11% |
16% |
| NBI (Index) |
3% |
14% |
Company Overview
NurExone is developing exosome-based therapies aimed at providing minimally invasive, highly targeted drug delivery, and regenerative medicine solutions. The company holds an exclusive, worldwide license from two leading Israeli universities for the development and commercialization of the technology. Under the licensing agreement, the company is required to pay 20.25% of revenue in royalties and licensing fees.
Founded in 2021 and based in Israel. Holds an exclusive worldwide license for the technology
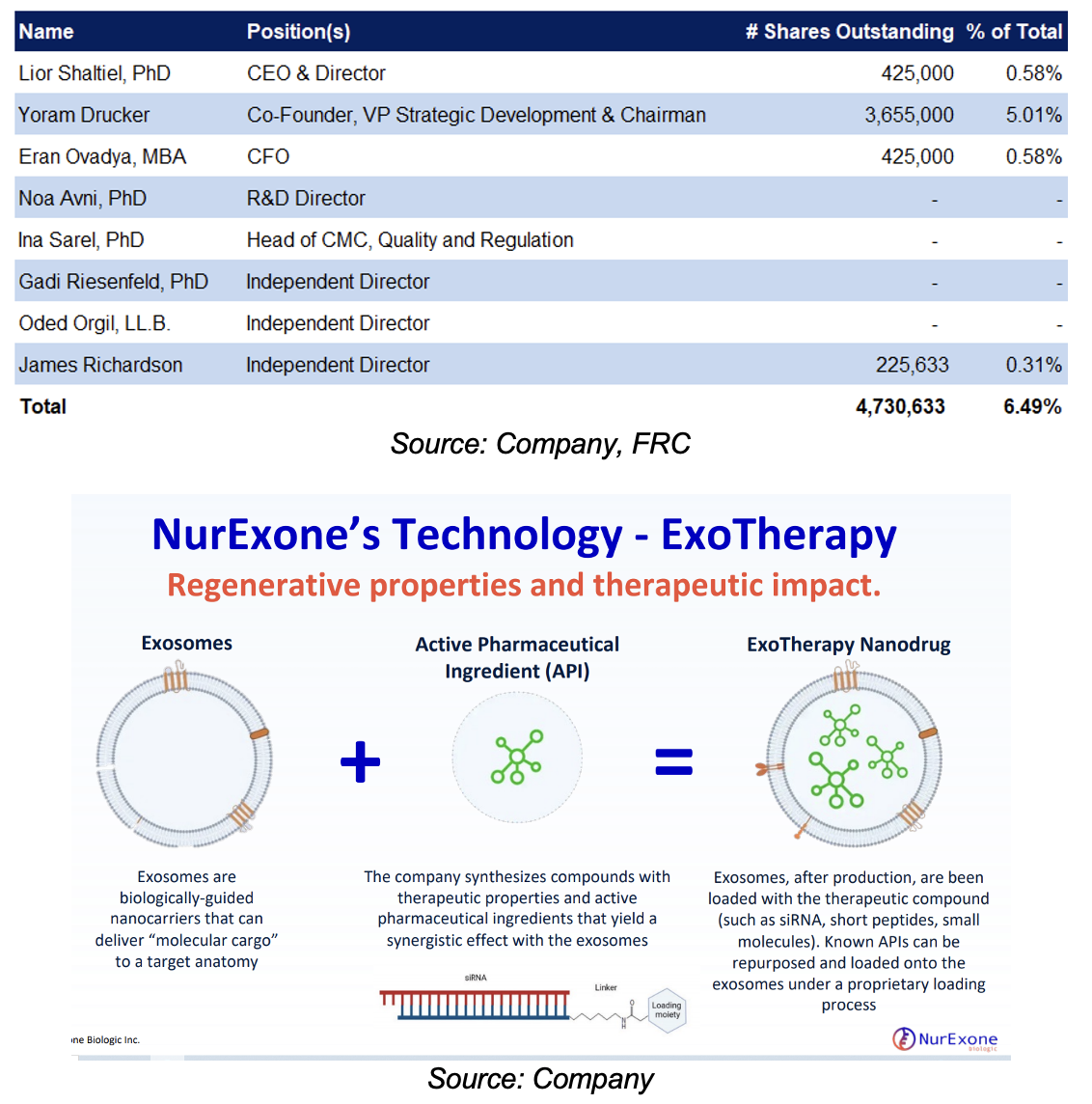
Established by experienced biotech entrepreneurs. Management owns 6.5% of NRX’s equity. Nine full-time and eight part-time employees
U.S. patent granted in 2023
Exosomes are nano-sized, membrane-bound vesicles (sacs) secreted by cells, and abundantly present in various body fluids, including blood, urine, saliva, semen, vaginal fluid, and breast milk. They play a pivotal role in intercellular communication, facilitating the transfer of vital biological molecules, such as DNA, RNA, and proteins, between cells. Various sources suggest that exosomes possess significant therapeutic potential, to serve as an effective, targeted drug delivery system. Exosomes' natural ability to target inflamed or damaged tissues, and their capacity to carry and deliver active pharmaceutical ingredients (APIs), make them a promising platform for targeted drug delivery and regenerative medicine.
In recent years, the exosome therapeutics and diagnostics industry has experienced significant growth, with over 50 companies actively engaged in R&D. However, there are currently no FDA-approved exosome products.
Exosome-based therapies are in early stages of development, aiming to improve upon existing gene and cell therapies
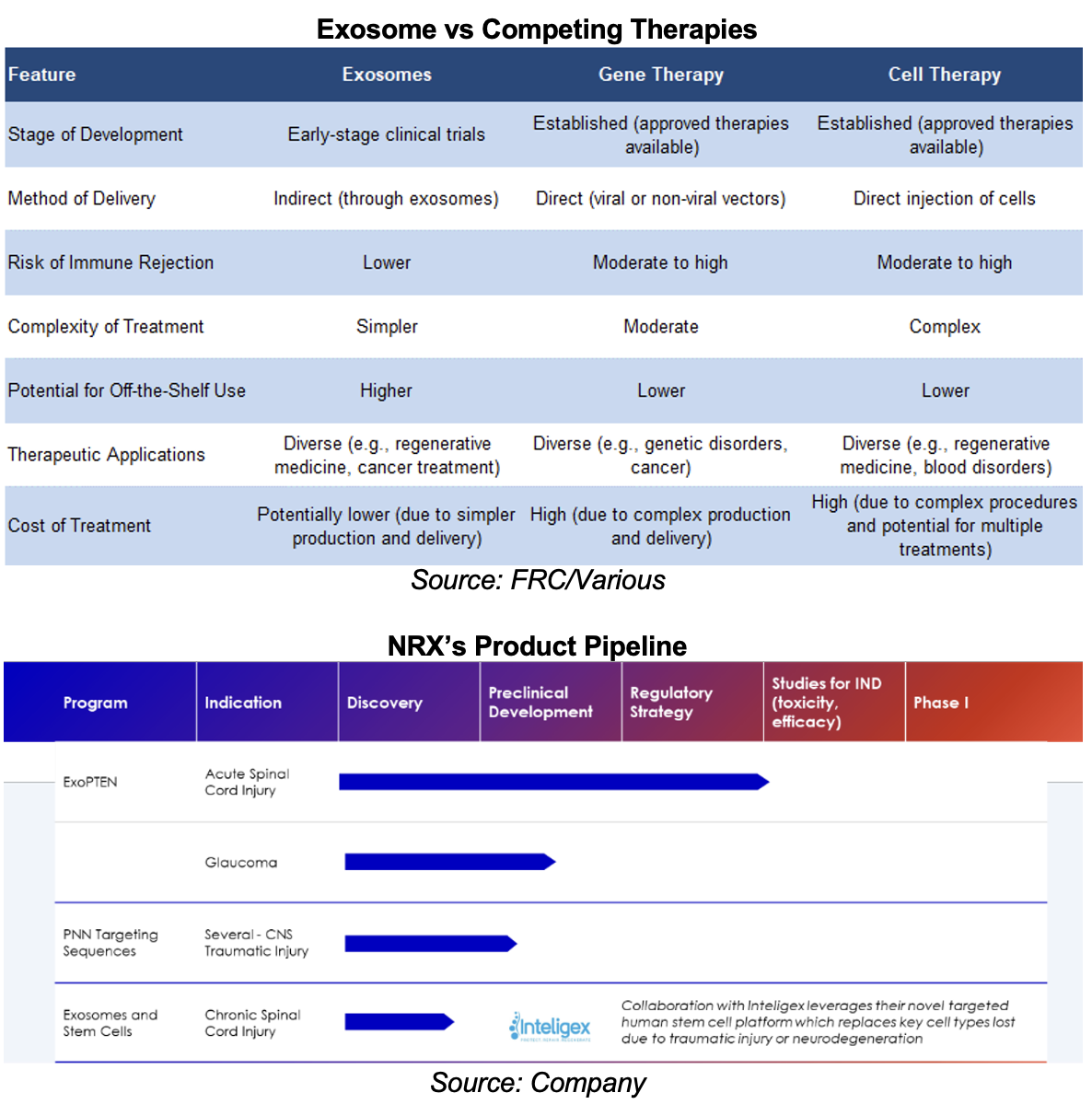
Studies indicate that exosomes offer unique advantages, such as reduced immune responses, targeted delivery, and the potential for off-the-shelf treatments
NRX is focusing on a broad range of applications within the central nervous system, including spinal cord injury (SCI), optic nerve injury, brain trauma, and various neurological disorders
NRX’s Lead Product - ExoPTEN
The company is developing ExoTherapy, an exosome-based therapy designed to stimulate neuroregeneration for the treatment of acute SCIs.
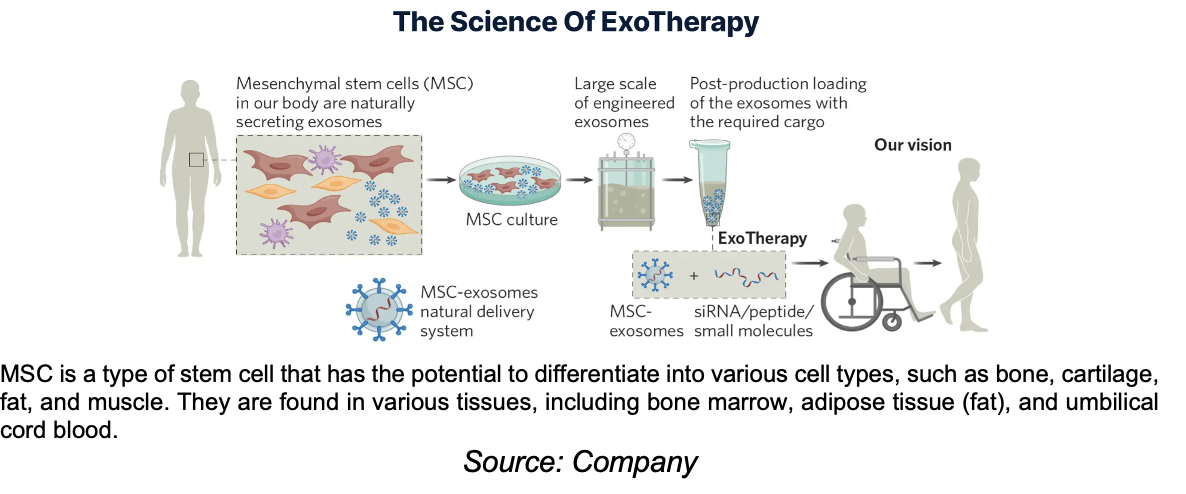
ExoPTEN utilizes exosomes loaded with a unique and proprietary special code (siRNA) as its active pharmaceutical ingredient
Management's goal is to develop ExoPTEN as a versatile treatment that can address a broad range of nerve injuries, including both acute SCIs, and optic nerve injuries. The technology has been validated in preclinical studies conducted on rats in 2018 and 2019. Key results include:
- Penetration of the Blood-Brain Barrier: Exosomes derived from MSC were shown to cross the blood-brain barrier, and migrate to the injured spinal cord region.
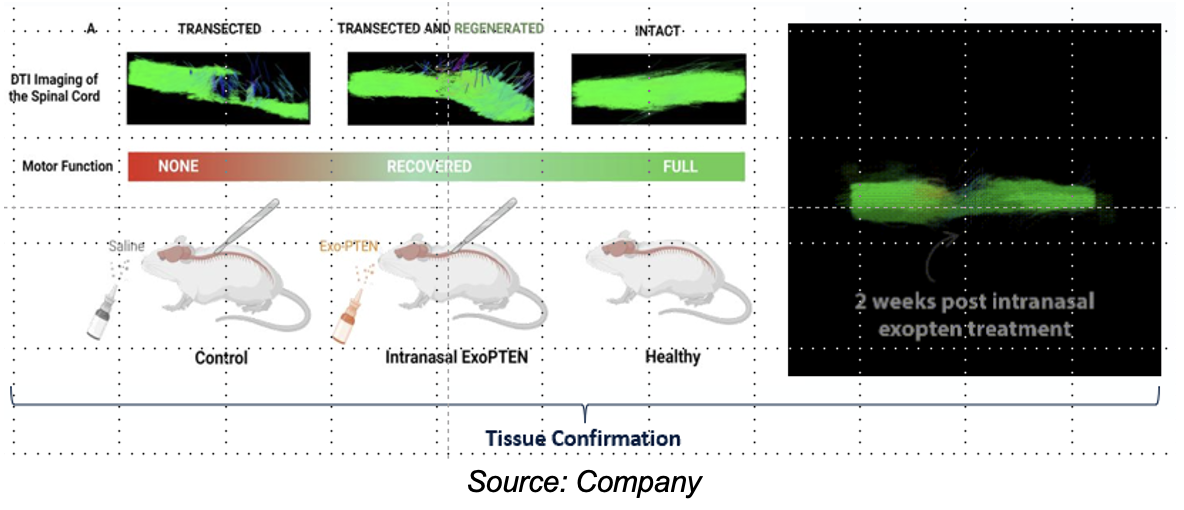
- Promising Results: ExoPTEN significantly improved motor function, sensory recovery, and urinary reflex restoration. Over 75% of laboratory rats treated with ExoPTEN recovered motor function. In cases of complete spinal cord lesions, some rats were able to walk again.
- Neuroregeneration: The study demonstrated the ability of ExoPTEN to promote the formation of new neural connections, partially repairing the damaged spinal cord.
- Off-the-Shelf Potential: Initial findings suggest that ExoPTEN may have the potential to be developed as a readily available therapeutic option.
Intranasal administration of MSC-derived exosomes loaded with siRNA-PTEN (ExoPTEN) was presented to rats with complete spinal cord lesions, resulting in significant functional recovery
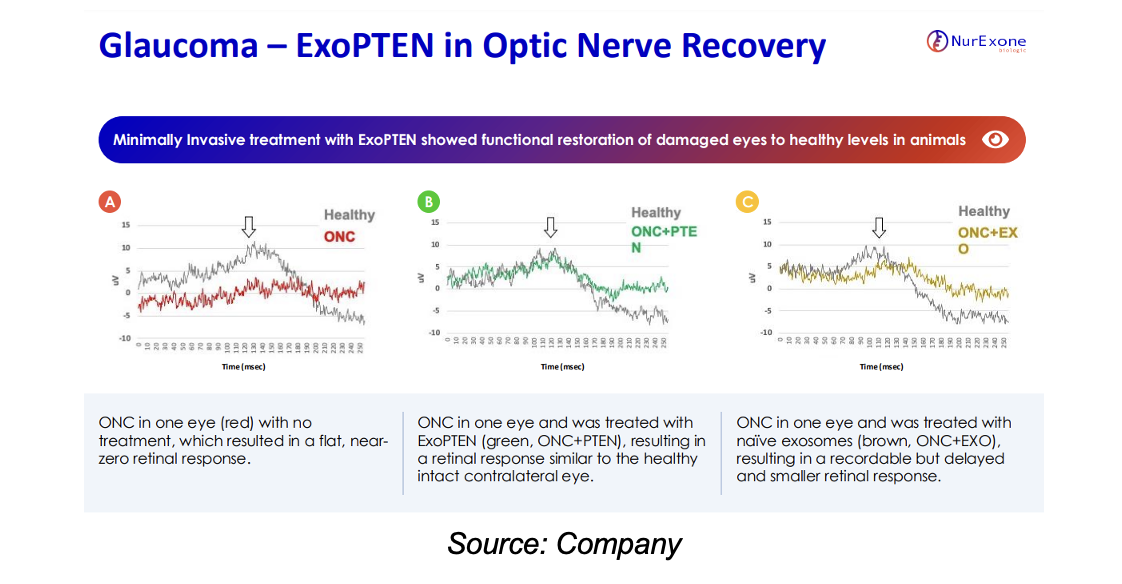
NurExone is also exploring the potential for treating glaucoma. Preclinical studies have demonstrated promising results in restoring vision following optic nerve damage—a key characteristic of glaucoma. Current treatments are primarily focused on preventing further damage, with limited options for regenerating or repairing damaged nerves.
We believe the expansion into the glaucoma market could significantly enhance NurExone's therapeutic portfolio, and address a critical unmet medical need
Upcoming Tests/Catalysts
ExoPTEN has received an Orphan Drug Designation from both the FDA and the EMA, potentially accelerating development and approval
Drug Development Pathway
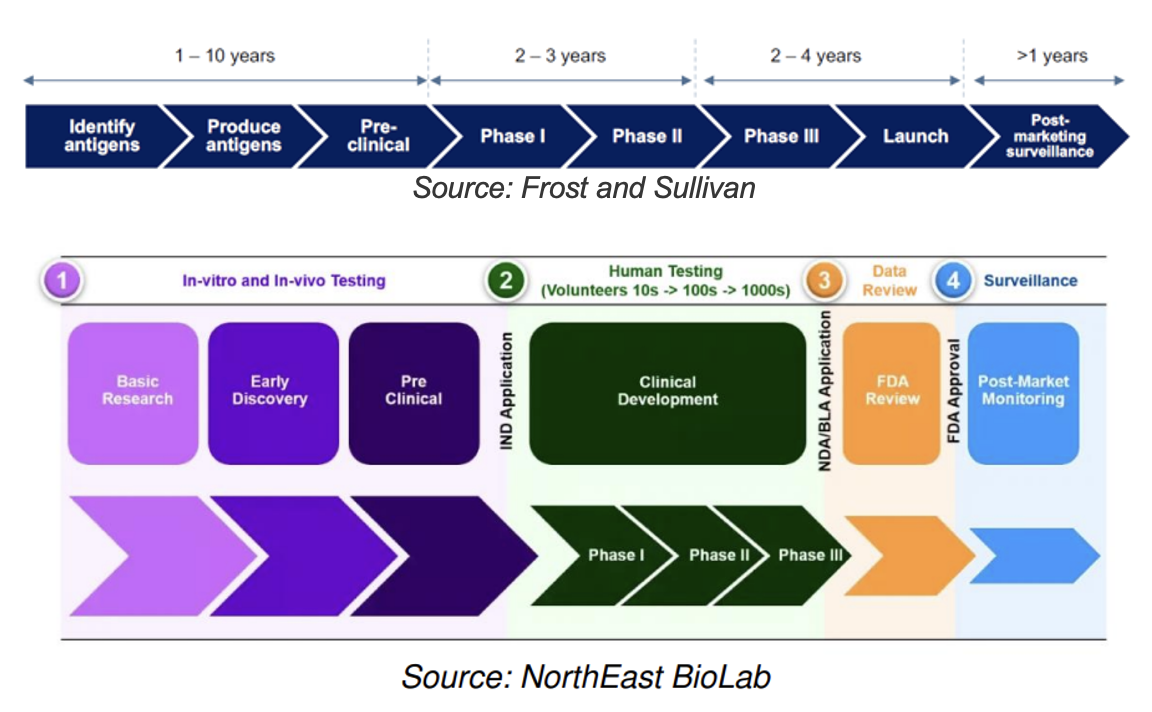
Historically, 75% of drug candidates have moved from phase I to II, 50% from II to III, 59% from III to approval, and 88% of those have received final approval (Source: National Library of Medicine), implying that 19% of candidates have advanced from phase I to approval.
NRX has also conducted a pre-IND meeting with the FDA, necessary for starting human clinical trials
The company expects to start phase one clinical trials by the end of 2025.
Market Potential
SCIs are complex conditions caused by trauma, such as motor vehicle crashes and falls, or non-traumatic factors like malignancy and degeneration, leading to severe morbidity and permanent disability.
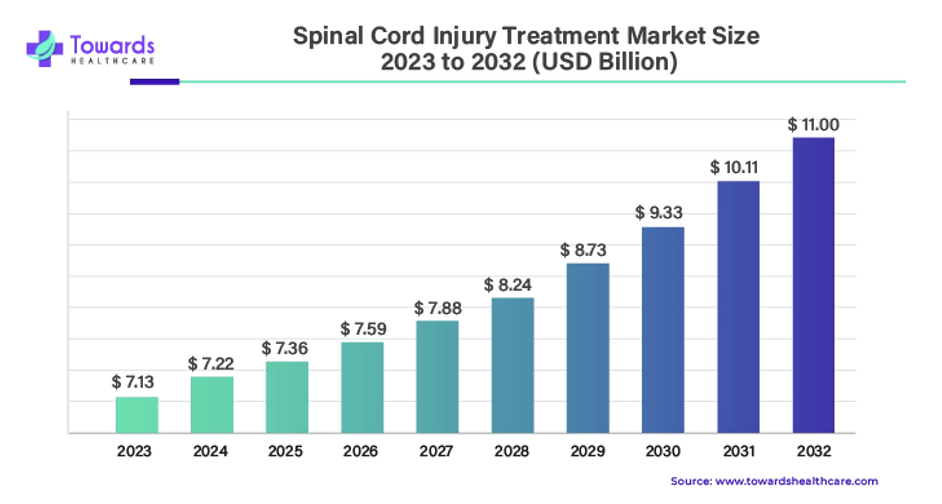
It is estimated that the global SCI treatment market will grow from $7.1B in 2023, to $11B by 2032, reflecting a CAGR of 4.8%
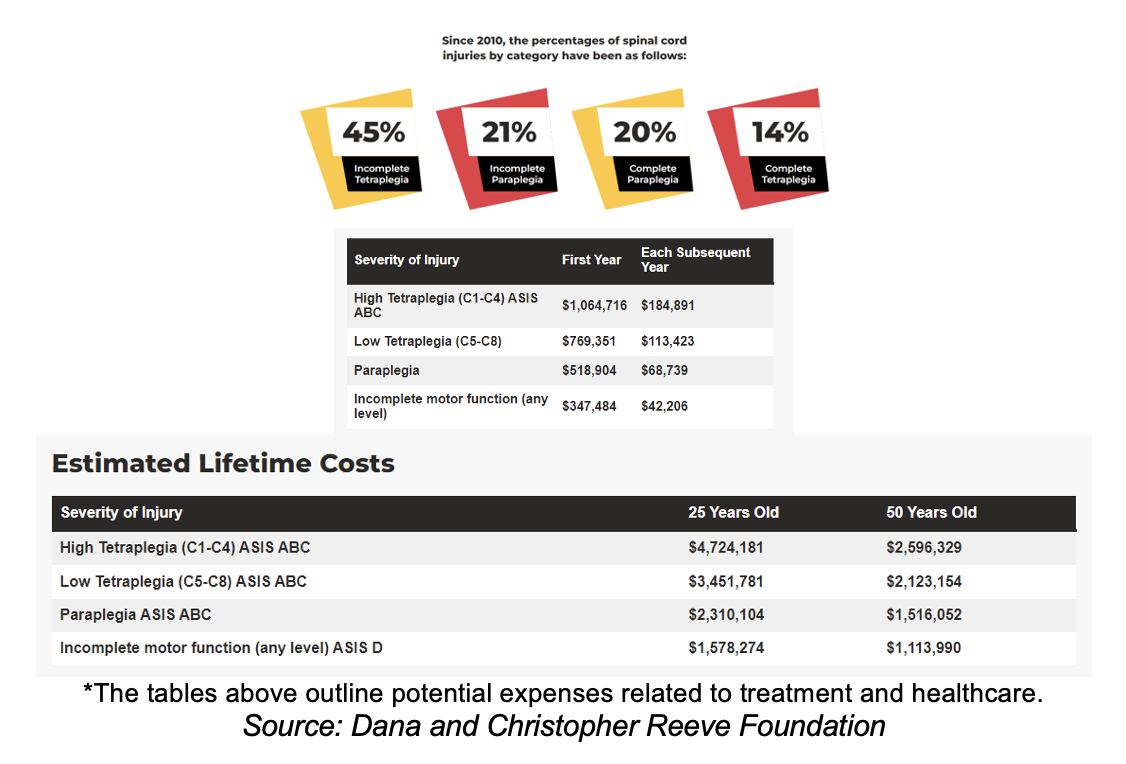
The Dana and Christopher Reeve Foundation reports that treating SCI can be extremely costly, especially for severe cases like high tetraplegia. In addition to substantial initial costs, ongoing care and rehabilitation expenses remain significant throughout a patient's lifetime.
Less than 1% of affected people experience complete neurological recovery by the time of hospital discharge
The National Library of Medicine reports that 250k-500k patients globally suffer from SCIs each year. In the U.S., about 17k new cases arise annually, with an estimated 282k people living with SCIs.
Globally, 40-80 people per million experience SCI annually (Source: WHO)
Approximately 80 million people worldwide are currently living with glaucoma
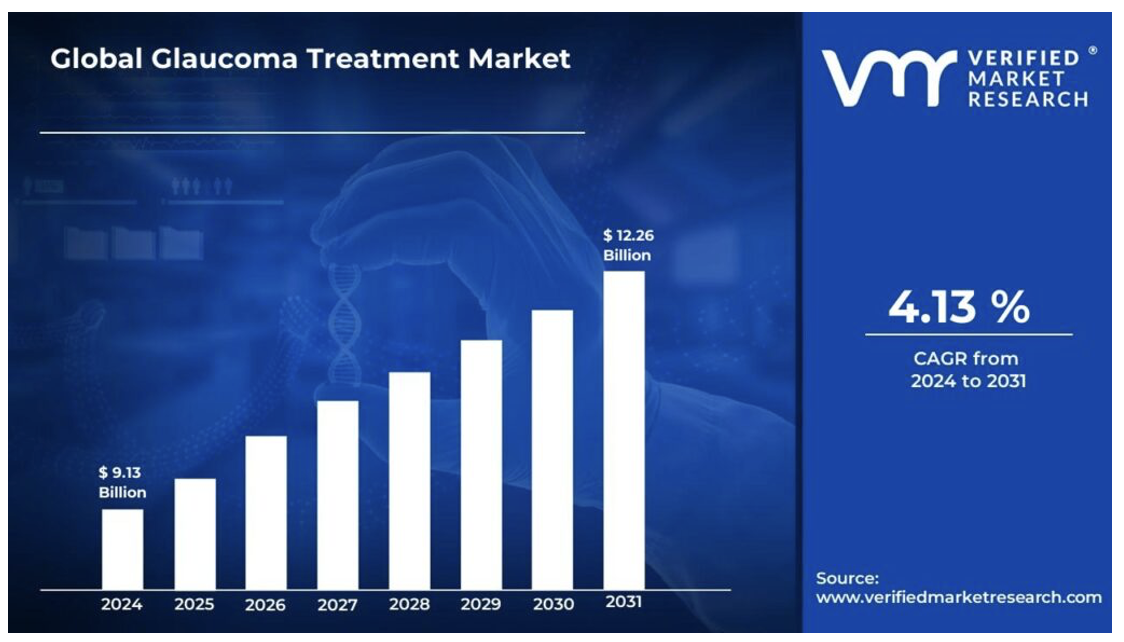
It is estimated that the global glaucoma treatment market will grow from $9.1B in 2024, to $12.3B by 2031, reflecting a CAGR of 4.1%, driven by an aging population, and increasing awareness and screening program
Financials
In pre-revenue stage. Current monthly burn rate : $400k/month. Raised $15M since inception. In-the-money options and warrants can generate up to C$9.2M; therefore, we do not anticipate any equity financings in the near term
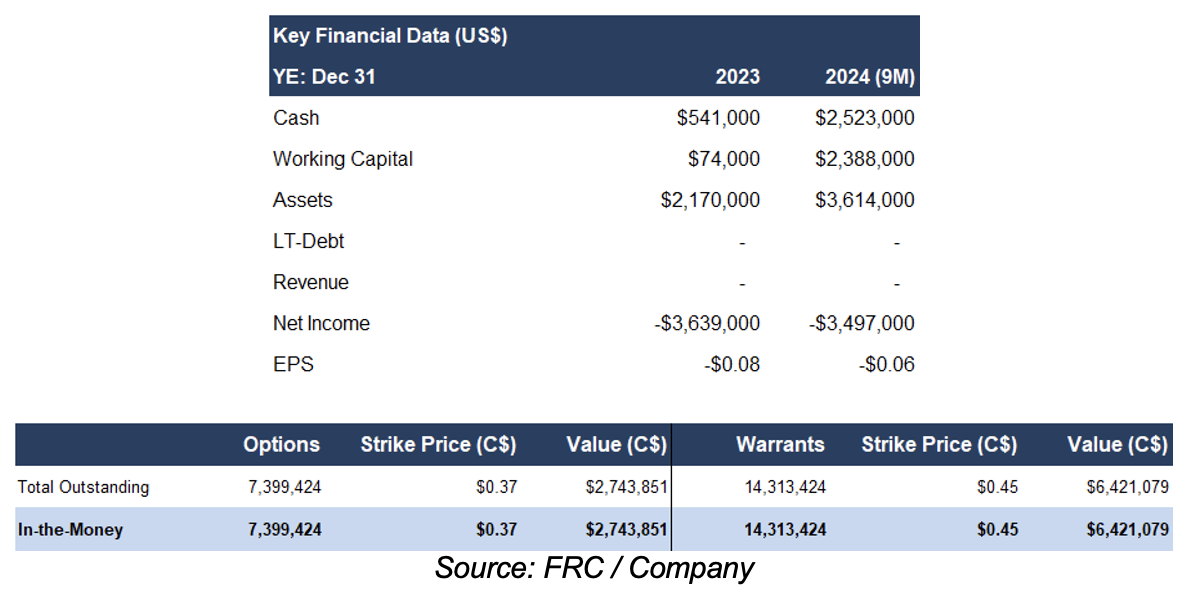
FRC Projections and Valuation
The following table lists major M&A deals in cell therapy and related fields.
Majors have acquired leading treatments for billions of dollars
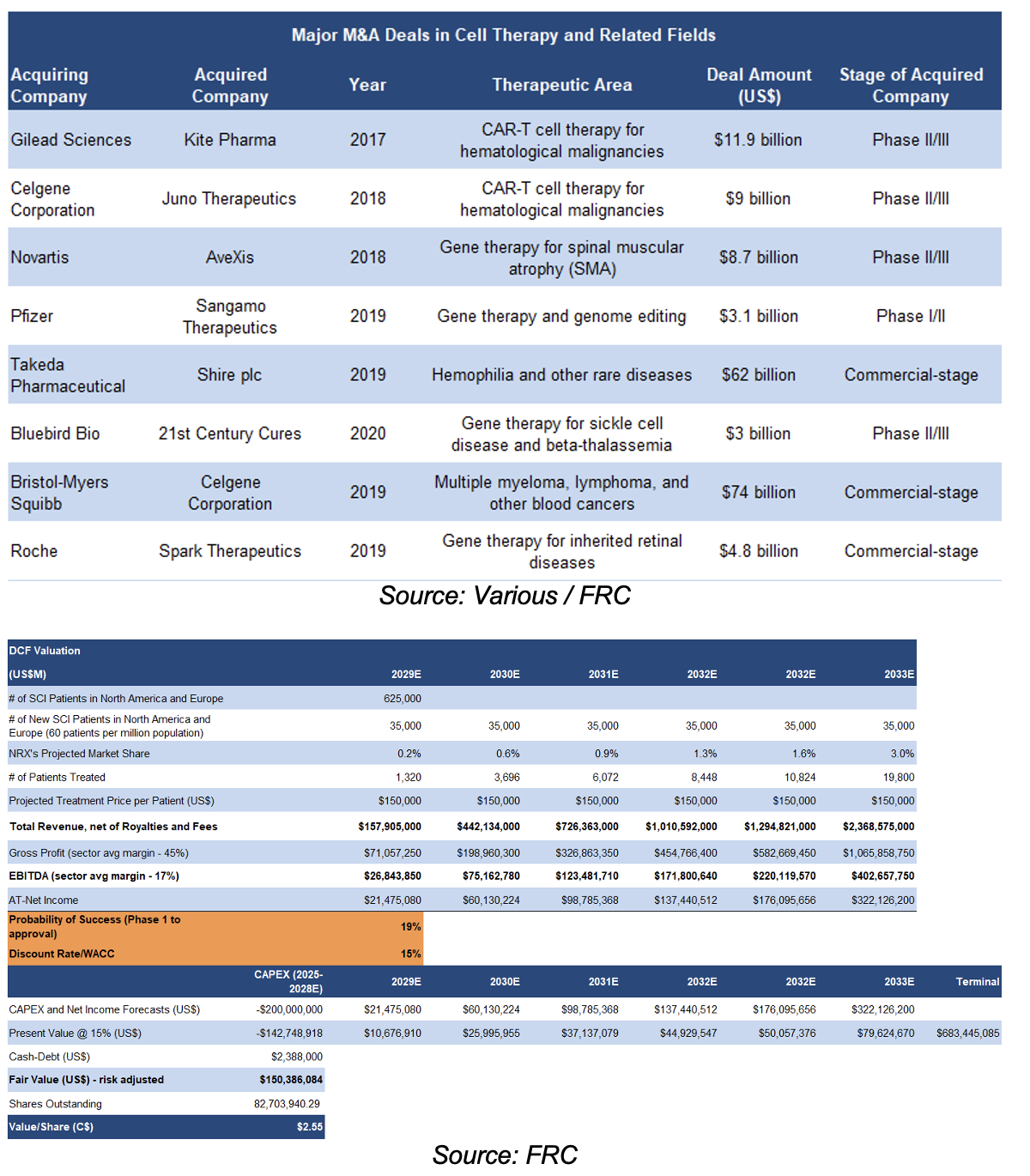
Our DCF model is based on the assumption that NRX will capture 3% of the SCI treatment market in North America, and Western Europe, by the sixth year of commercialization
Modelling $200M in CAPEX for advancing towards commercialization
We arrived at a DCF valuation of C$2.55/share
Key Assumptions:
- We are applying a probability of success of 19%, consistent with the sector's average success rate.
- We are using a relatively high discount rate of 15%; our typical range for valuing pre-revenue companies is 10% - 15%.
- We are assuming an average treatment price of $150K, which aligns with that of conventional cell therapies and orphan drugs. Note that this price is significantly lower than the current treatment options for SCI, as shown in the tables presented earlier in this report.
- Sector-average EBITDA margins have been applied.
- For conservatism, we are not accounting for any value from the company’s other target applications.

We are initiating coverage with a BUY rating, and a fair value estimate of C$2.61 per share (the average of our DCF and real options valuations). While NRX faces the inherent risks of an R&D-focused biotech, and exosome therapy remains a nascent field with no FDA-approved products, we view it as an innovative player in developing treatments for neurological disorders. Its lead candidate, ExoPTEN, has shown encouraging preclinical results, and received an Orphan Drug Designation. The typical exit strategy for pharma and biotech companies is acquisition by larger firms upon the successful completion of promising clinical trials.
Our real options valuation is C$2.67/share
We believe a real options valuation model is valid when valuing development-stage biotech companies, as the model takes into account management’s ability to pursue, abandon, or delay drug development
Risks
As with all R&D stage biotech companies, we are assigning a risk rating of 5 (Highly Speculative
We believe the company is exposed to the following key risks (not exhaustive):
- Limited operating history
- In pre-revenue stage
- No guarantee that any of its drugs/treatments will be commercialized
- Potential for delays in trials; unfavorable results
- Will need to pursue equity financings, implying potential for share dilution


















